New battery materials for a renewable energy future
Where will the next breakthrough technology in renewable energy come from?
Research at the European Spallation Source will help to relieve the energy storage bottleneck, one of the chief obstacles to renewable energy innovation.
A key characteristic of renewable energy is its variability. Solar energy is frequently obscured or unavailable, waves come in waves, and the wind is famously fickle. Uninterrupted energy flow requires the storage of energy to compensate for this variability. The coupling of energy storage to renewable energy generation, however, is still in its infancy.
Among the different energy storage technologies, lithium-ion batteries hold great promise. Though widely used commercially in consumer electronics and electric vehicles, it is understood that their application could be significantly expanded through innovation. Problems with performance, cost, safety and scalability can be traced to an insufficient understanding of the materials and processes of these batteries at the molecular and atomic scales.
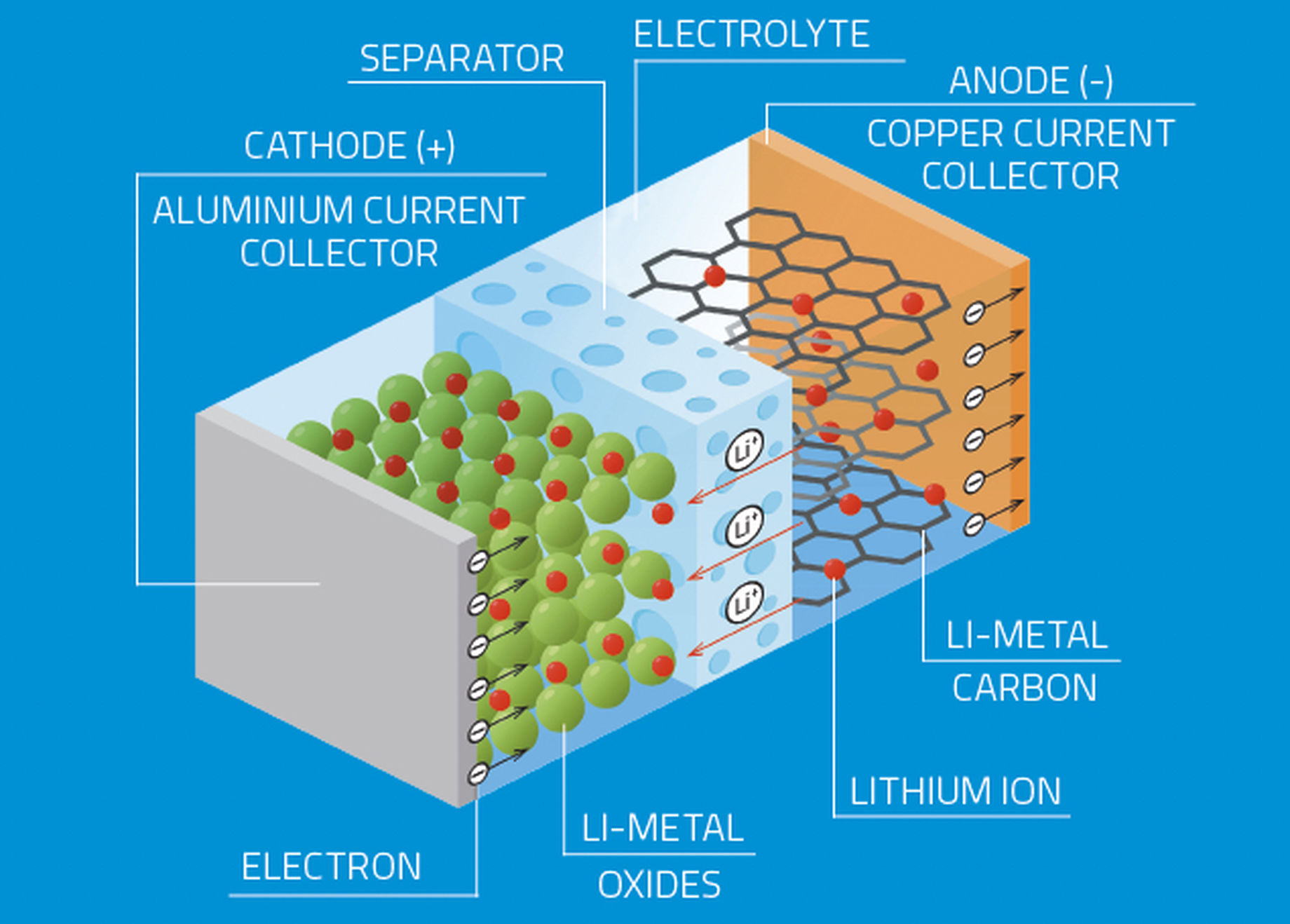
A lithium-ion battery is a type of rechargeable battery in which lithium ions move from a negatively charged electrode to a positively charged one during discharge, and back again when charging. This flow occurs at the atomic scale and is therefore difficult to observe and model, limiting researchers’ ability to experiment with new materials.
Meanwhile, those materials, used for the electrodes and the electrolytes of a lithium-ion battery, largely determine its capabilities. An in-depth understanding of material behaviours within this complex electrochemical environment is thus critical for the development of advanced materials that will lead to better batteries.
These advances are our best hope for renewable energy storage solutions grand enough to contribute to the transformation of the global energy sector.
Lithium-ion batteries currently in development include nickel, manganese or cobalt compounds that together with increased lithium content have combined to steadily increase storage capacity by increasing the battery’s energy density. However, the batteries engineered with these experimental materials are not sufficient to meet the safety, durability and performance needs of transformative technologies. These are optimisations that will primarily be achieved by exploring new materials using neutrons.
What will we see at ESS?
Neutrons are the perfect tool for studying positive electrodes with a chemical composition based on nickel, manganese and cobalt. Due to their very similar atomic numbers, which place them near one another on the Periodic Table, these three metals appear indistinguishable when probed with X-ray light. With neutron scattering, however, researchers are able to see clear differences in the three. Astonishingly, neutrons can also locate the position of lithium ions in the structure, and help to visualise the lithium pathways.
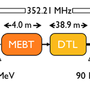
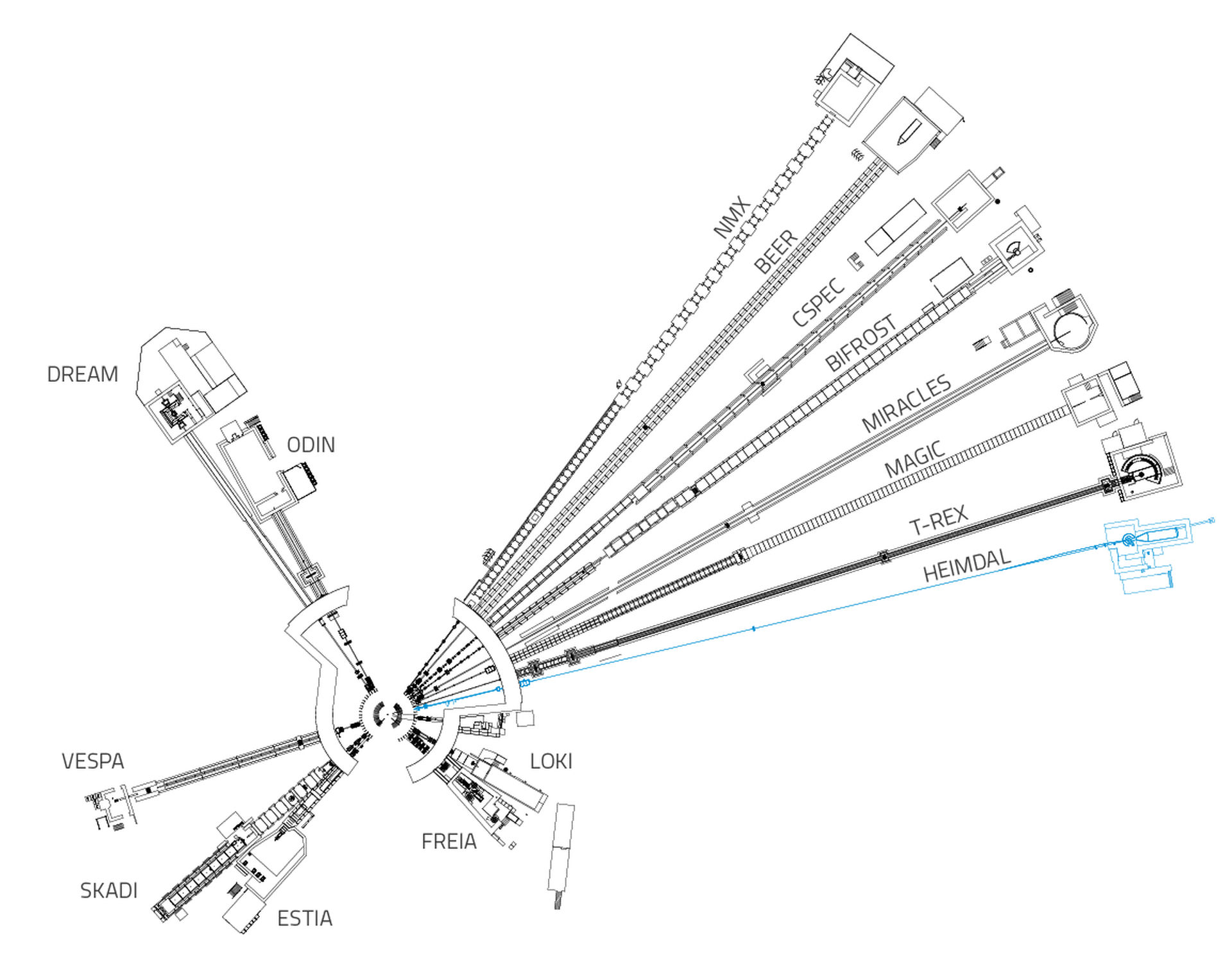
At ESS, critically, the HEIMDAL diffractometer will also enable the study of experimental battery technologies in real time and under real-world operating conditions. It is HEIMDAL’s innovative design that enables this breakthrough. The instrument combines two highly complementary neutron methods: powder diffraction and small-angle neutron scattering. Coupled to the unparalleled brightness of the ESS neutron source, this will allow for a uniquely broad range of experiments within a single instrument setup.
HEIMDAL, for the first time, will reveal a clear and detailed picture of an entire battery cell, at multiple length scales, during operation. This will include the structure and dynamics of its electrodes and electrolytes, as well as the behaviour of its mobile ions, all interacting while the battery is charging or discharging.
Researchers will be able to swap out nickel, manganese and cobalt components as required for each investigation, and observe the impact of the many possible variations on overall battery performance and safety. This will provide an unparalleled understanding of a lithium-ion battery’s fundamental processes, while also significantly speeding up the development of this technology so critical to the transformation of the planet’s energy profile.
Beyond renewables
Rechargeable lithium-ion batteries have already had a huge impact on society, enabling a mobile, digitally connected lifestyle by powering laptops, smartphones, electric wheelchairs and more. They are also essential to military, electric vehicle and aerospace applications, industries that have been transformed by advances in battery materials.
Due to its uniquely versatile design, HEIMDAL will revolutionise the study and development of advanced functional materials used across a wide range of complex systems. In addition to energy-related materials, examples include the study of multiferroics for use in IT applications, the development of safer hydrogen storage materials, the manufacturing of more robust magnets for trains, investigations into the many potential uses of nanoparticles, and the pursuit of viable carbon sequestration systems.
HEIMDAL
HEIMDAL is a multi-length-scale neutron scattering instrument designed for studying advanced functional materials. Specialities include: energy-related materials, composites, scaffolds, phase transition and nucleation, and materials with magnetic properties.
Fifteen instruments are currently under construction at ESS.
The in-kind partner institutions collaborating with ESS to design and build HEIMDAL are:
This feature is part of an ongoing Future Science series; a set of stories highlighting some of the research opportunities that will be made possible by instruments currently under construction at ESS.