Bringing the most effective cancer drugs to market sooner
How will innovative molecular mapping help the fight against cancer?
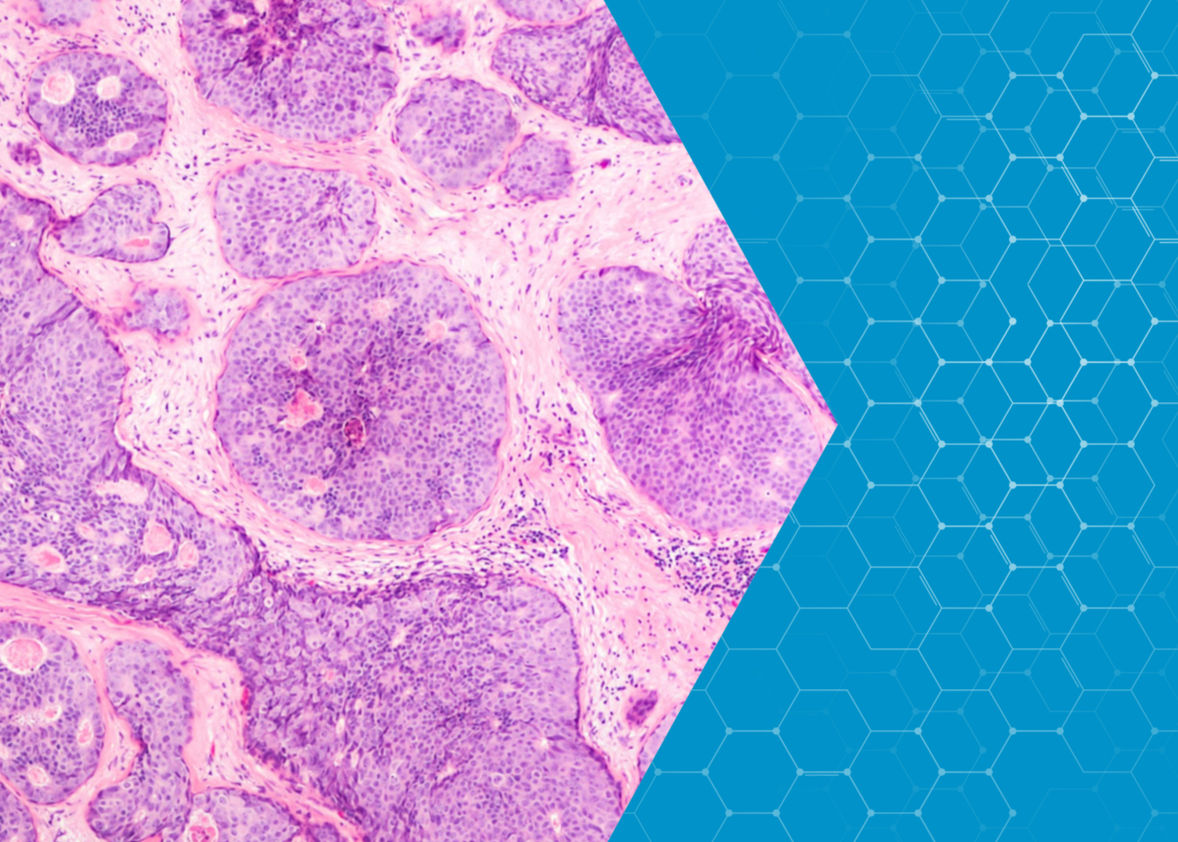
Fundamental to defeating cancer is the ability to map and describe tumours at the molecular level. Research at the European Spallation Source will help to chart tumour cells in unprecedented detail, providing the basis for a new standard of cancer diagnosis and treatment.
Current cancer drugs are hunting their prey in a dark forest, and with no map. As a result, they don’t always find their target or shoot straight when they do.
To design cancer drugs that can target tumours more precisely and with greater success, it is critical to map the architecture of tumour cells at the molecular level. To do this, researchers must shine a bright light on the diseased tissues that make up this dense, well-protected forest to capture the lay of the land.
Such work is already in progress, but the clock is ticking. The creation of molecular maps for tumour cells will open up cancer drug studies to innovation currently not possible. It will enable the identification of vulnerabilities that can be targeted by treatments, while providing new insight into metastasis and other important processes in tumour development.
Developing such maps is, of course, complex. Beyond revealing the size and shape of the tumour, mapping the individual fats, proteins and water molecules in the tumour’s cells will be needed for a real step-change in drug design. The state of water in these cells turns out to be a particularly strong indicator of cancer status and progression.
Water molecules have a strong electrical polarity, meaning that there is a strong positive charge on one side of the molecule and a strong negative charge on the other. This causes water molecules to bond to each other and to other charged surfaces. Researchers have observed that in specific breast cancer cells this bonding mechanism may be different from that seen in healthy ones.
Connecting the mobility of water molecules to cancer behaviour is a promising development. The growth or remission of a tumour can be manifested by changes in the functioning of its water molecules, while new therapeutic treatments could also alter the structural organization of the confined water in the cells.
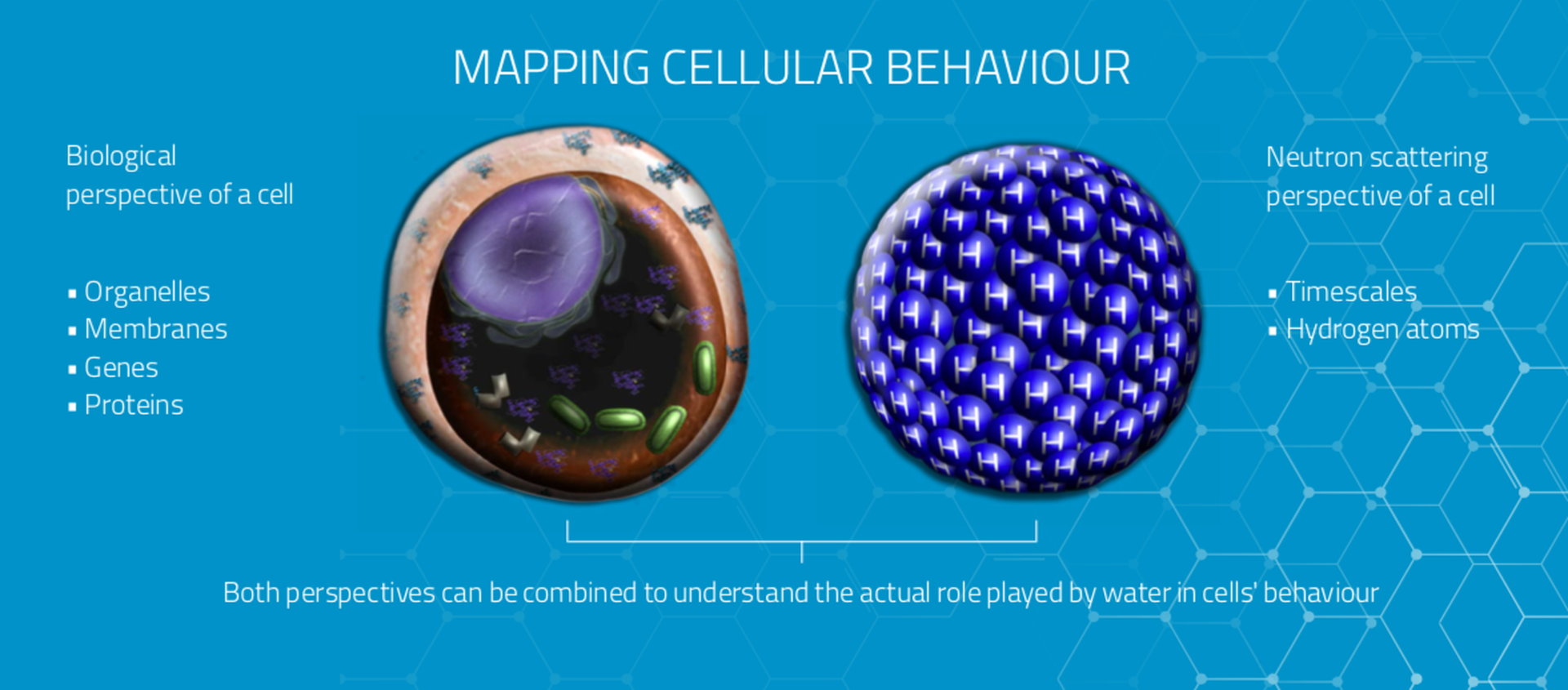
It is currently possible to look at whole cells and discern the state of water—bound or unbound to cellular components—but if researchers hope to relate changes in water behaviour to pharmaceuticals they will need better tools. These tools will need to record more data on different types of cells at a faster rate. The molecular maps resulting from such work would enable drugs designed to interrupt disease progress more effectively. They will also accelerate the drug development process, resulting in a quicker pathway from the laboratory to the patient.
What will we see at ESS?
The unparalleled brightness of the ESS neutron source will give researchers one of the new tools essential to advance this research. Only neutrons can give us temporal and spatial information at the same time, providing the complex measurements needed to investigate the structure and dynamics of water in cells.
Using different neutron scattering techniques, researchers can monitor the motions of water at a wide range of time scales. This enables a huge body of data to be collected on the state of water molecules at any given moment. The neutrons at ESS will also enable such measurements across length scales ranging from whole tissues all the way down to the structures of the proteins within a single cell.
Current studies made across such a broad range of parameters require an impractical matrix of access to different techniques on different instruments and research sites across the globe. The MIRACLES spectrometer at ESS will be the only neutron instrument in the world to offer a parametric study approach. Designed to assume the value of all different factors at play during cellular interactions, MIRACLES will greatly reduce the length of time needed to conduct experiments and to collect and analyse the data.
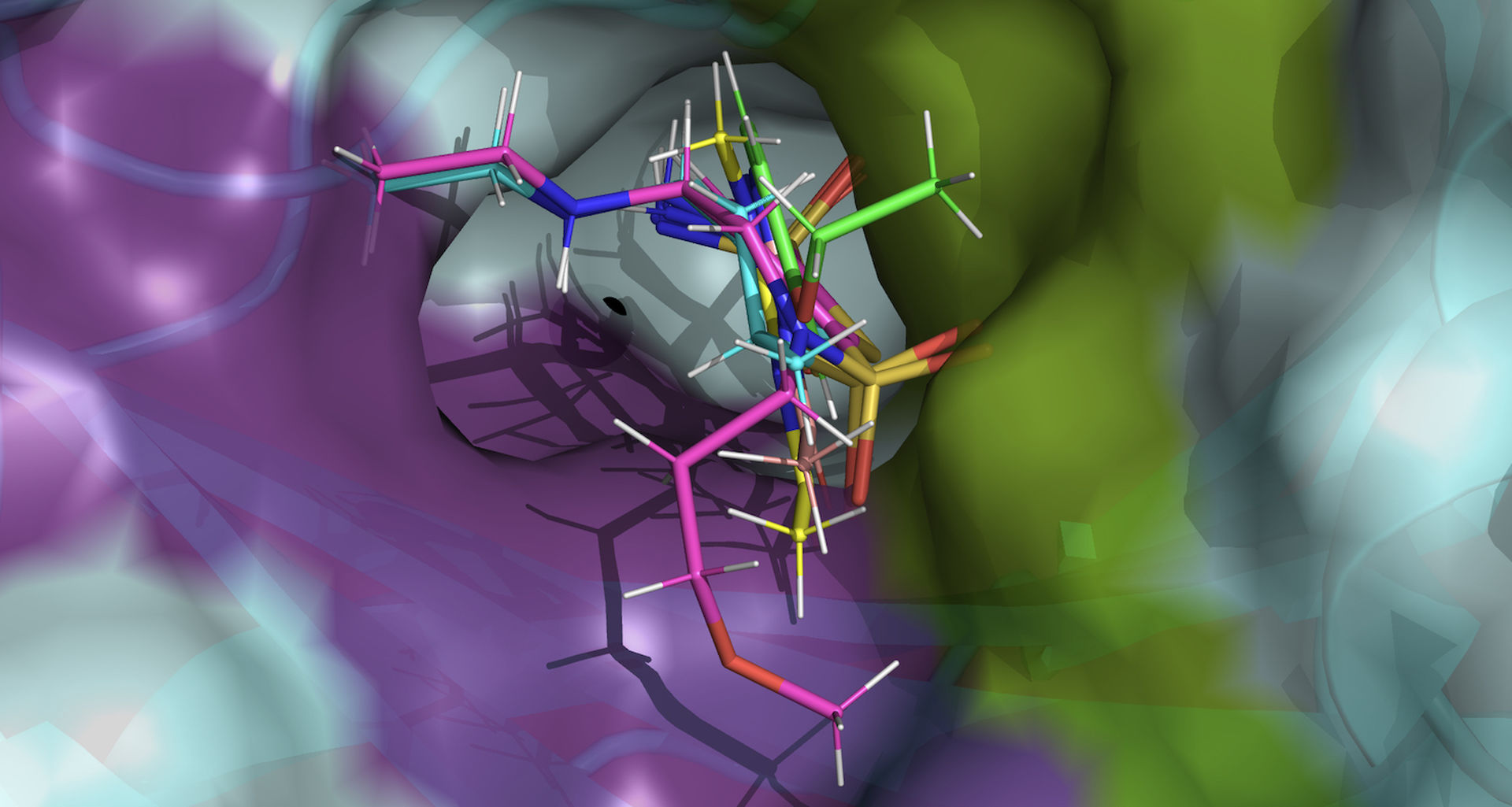
For the first time, researchers using MIRACLES will be able to evaluate whole cancerous tissue samples and observe all hydrogen atoms in the tissue’s water molecules simultaneously. It can conduct these analyses across many types of the most common cancer cells. Able to observe dynamic processes over a wide energy range, MIRACLES will also enable researchers to observe the interactions between prospective drugs and their molecular targets all on a single instrument.
The construction of MIRACLES additionally includes the optimisation of software needed to analyse the instrument’s data more quickly and accurately. Molecular biologists and drug developers will therefore be able to access the experimental data from MIRACLES and apply it to ongoing studies, bringing the best drugs to market sooner than with current processes.
Beyond cancer
The unique application of MIRACLES to cellular biology will have implications for the study of other diseases
Additionally, the ESS spectrometer’s outstanding flexibility and ability to probe magnetism will contribute to innovation across the energy, climate and materials science sectors. MIRACLES will be a particularly exciting new partner in the wide-open field of polymer science. These remarkable materials have shown promise in dozens of innovative applications, and are being investigated for their potential in the urgent quest to capture greenhouse gases in the fight against climate change.
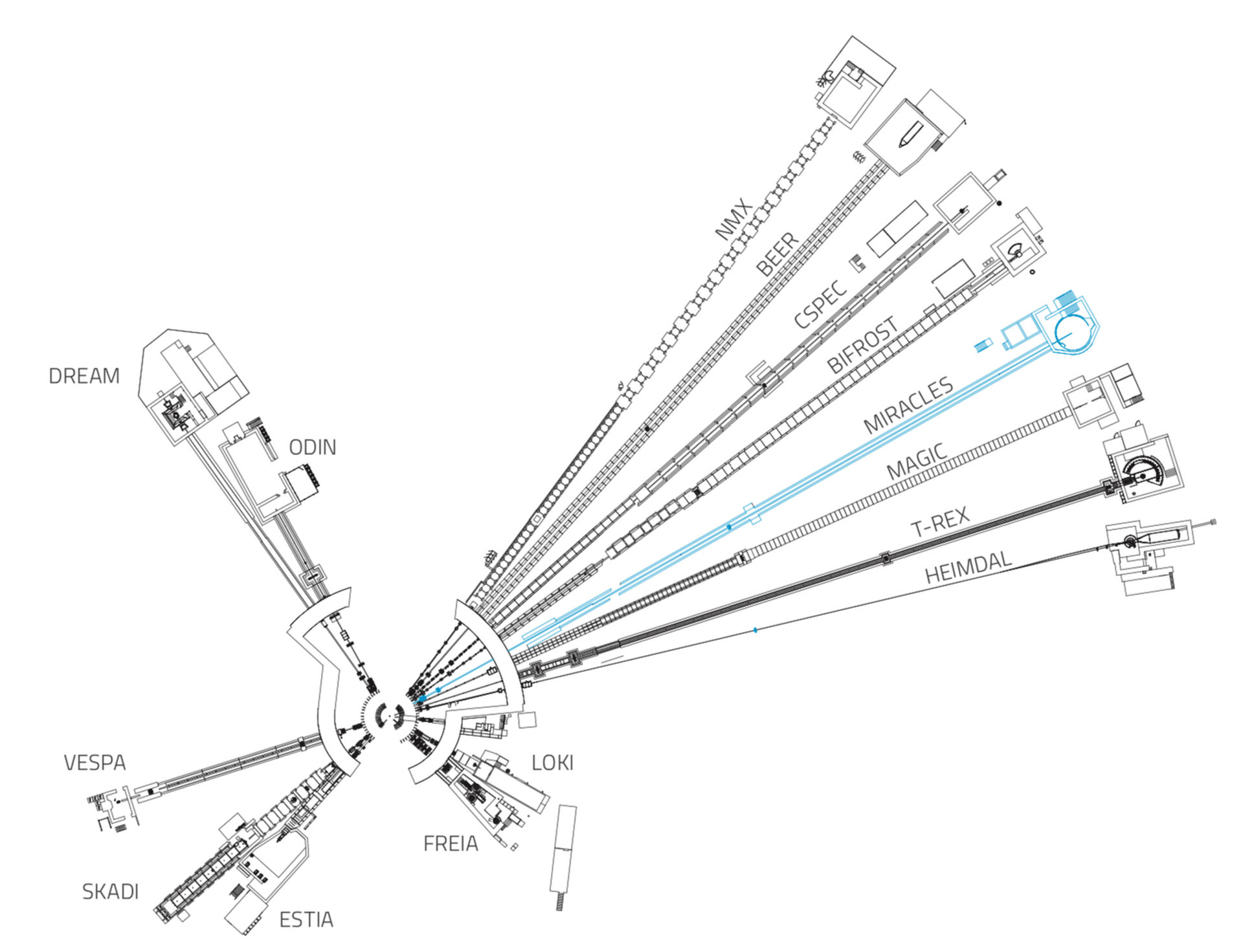
MIRACLES
MIRACLES is a time-of-flight backscattering spectrometer. Revealing dynamic processes over a wide energy range, it will serve life science, polymer science, energy materials, magnetism studies and much more.
Fifteen instruments are currently under construction at ESS.
The in-kind partner institutions collaborating with ESS to design and build MIRACLES are:
This feature is part of an ongoing Future Science series; a set of stories highlighting some of the research opportunities that will be made possible by instruments currently under construction at ESS.