New tools in the fight against air pollution and global warming
How can we engineer metal-organic frameworks to capture and neutralise harmful pollutants?
Research at the European Spallation Source will probe the underlying chemical reactions in catalytic systems, enabling them to be optimised to improve air quality and reduce greenhouse gases.
Air pollution occurs when harmful or excessive quantities of substances including gases, particulates and biological molecules are introduced into Earth's atmosphere. The term not only applies to “bad air” but includes the chemicals that degrade the ozone layer and the greenhouse gases that cause global warming.
Poor air quality also compromises the air we breathe, degrading health and well-being across the board, and its impacts are growing. The World Health Organisation estimates that every year about seven million people die from pneumonia, lung cancer and strokes directly linked to exposure to the fine particles in polluted air. In some parts of the world air pollution is now the leading cause of premature death.
The most common pollutants are the result of human activities. Direct emissions from automobiles and industry produce the most harmful and plentiful of these, including carbon, sulphur and nitrogen oxides. The very industries that drive the world economy are now on a path that threatens its viability.
Science and industry are working together to reverse this trend by developing materials that promise a more controlled application of the catalysis of individual chemical reactions. Catalysis is the process of accelerating a desired chemical reaction by adding a small amount of a substance—the catalyst—that is not consumed by the reaction and therefore can continue to perform this task repeatedly.
Catalytic converters and particle filters in automobiles, as well as scrubbers for smokestacks, have revolutionised the fight against air pollution by capturing and converting polluting particles and gases before they are released into the air. The solutions have not kept pace with the pollutants, however, and new materials are needed to bring catalytic technologies to the next level.
There is promising research demonstrating that a pioneering class of materials known as metal-organic frameworks (MOFs) could radically expand the application of capture, separation and catalysis in the fight against global warming and air pollution.
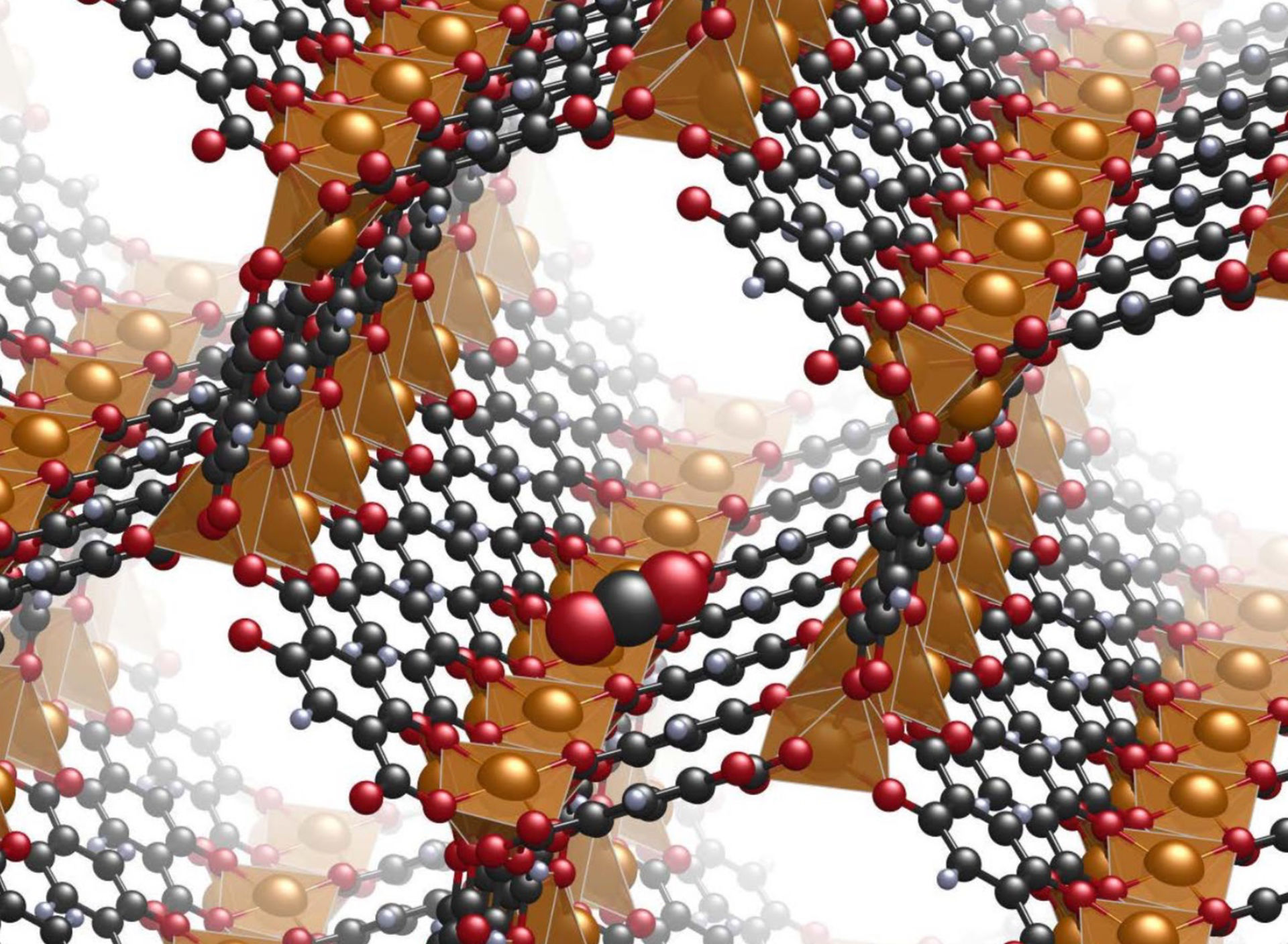
As the name implies, a MOF is a part-metal, part-organic crystalline material. It can form a one-, two-, or three- dimensional porous structure with an enormous surface area far out of proportion to its size. First synthesised less than 20 years ago, MOFs have been dubbed the “miracle material” because, theoretically, they can be engineered to serve a broad range of unique functions.
Some MOF structures, for instance, contain stable molecular-scale voids suitable as a site to catalyse polluting particles or store gases that otherwise would escape into the atmosphere. These molecular-scale lattices can in turn be organised into larger-scale porous materials, tailored to attract specific pollutants and lead them to the MOF trap. Such systems would allow for tunable, highly efficient cleansing of polluted air streams, for example from industrial exhaust.
A better understanding of the chemical reactions that take place within the pores of MOFs—and how those reactions affect the overall catalytic system—will aid in optimising them to select for the right size and shape of molecular compounds for storage, separation or conversion. This would enable MOFs to perform at the scale and sensitivity required to tackle a problem as large as air pollution.
What will we see at ESS?
Neutrons have no electric charge that might influence the processes they are probing, and at the same time can penetrate deeply inside the intriguing structures of MOFs with advantages no other analytical method can provide. Neutrons are particularly useful in tracking the movement of hydrogen and a handful of other atoms carried by the pollutants, allowing researchers to observe how the pollutants interact with the MOF. Neutrons, remarkably, can track each element as it progresses through the entire catalytic process.
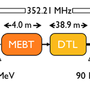
Higher neutron brightness means that these observations can be made in greater detail and while chemical reactions are in progress. The European Spallation Source will be the brightest neutron source on the planet, enabling its one-of-a-kind spectrometer VESPA to host experiments in catalysis and many other chemical, biological and physical processes that are not possible today.
VESPA will enable scientists to subject an operational MOF catalytic system to the ESS neutron beam in order to observe and monitor its behaviours from start to finish. By recording the reaction pathways of the various pollutants inside MOFs, VESPA will track which chemicals are converted, which are captured and stored, and which are released. This will help to identify which crystalline structure is best suited to capturing which pollutant so that MOF systems can fulfil their potential as particle killers.
Beyond air pollution
Metal-organic frameworks have the potential to transform several major industries. The instrument leverages a common technique known as vibrational spectroscopy that is used in research and industrial laboratories worldwide. VESPA will be the world-leading neutron vibrational spectrometer, and it will be used to explore an extremely wide range of applications in chemistry, physics and materials science.
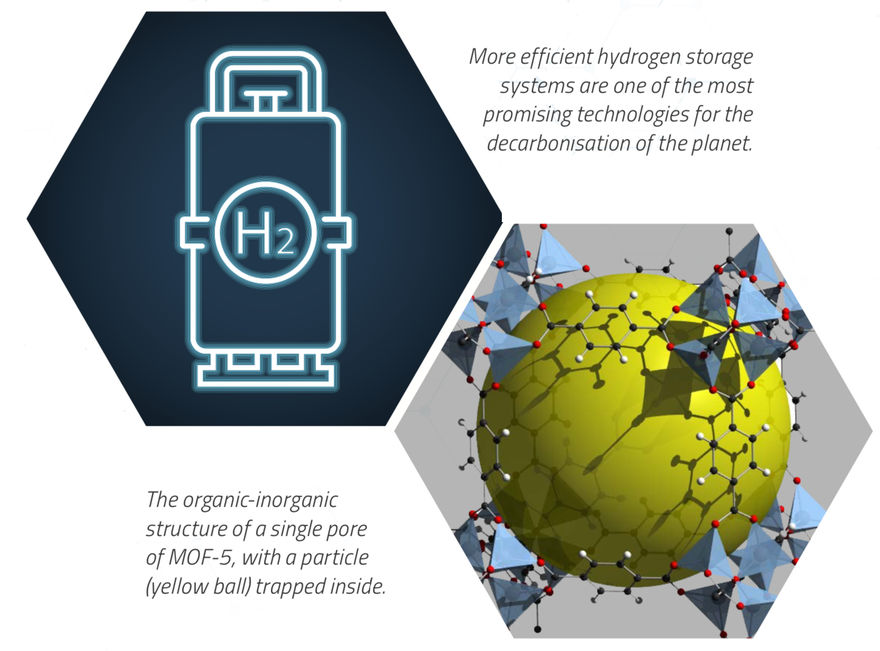
The instrument’s ability to track the activities of hydrogen atoms, carbon oxides and nitrogen oxides represents a huge scientific opportunity in many diverse areas where in situ measurements of catalysis are currently a challenge. Key applications for renewable energy technologies like hydrogen storage, better batteries and solar harvesting will be targeted, as well as continued investigations into game-changing polymer applications and the advancement of targeted drug delivery.
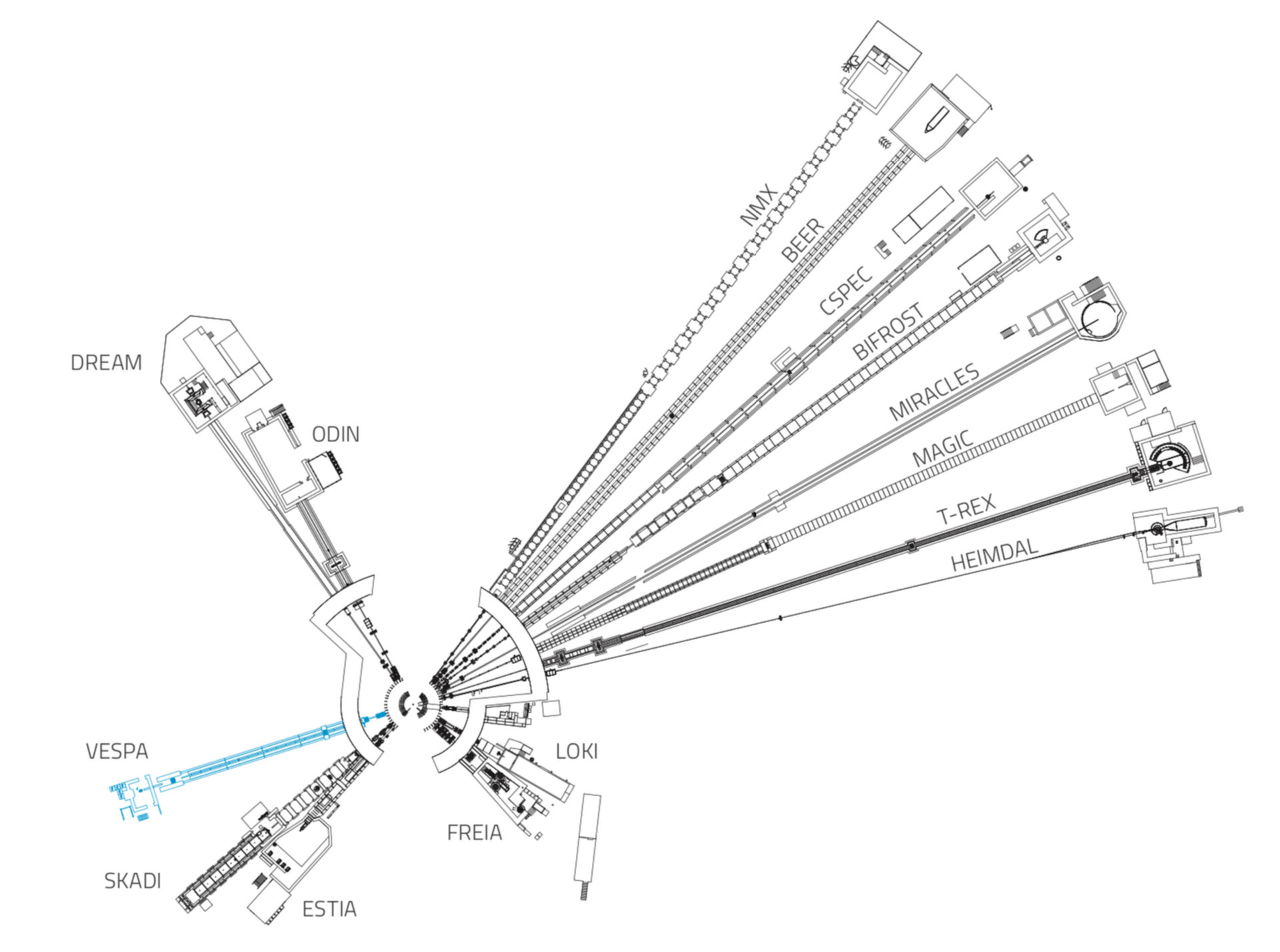
VESPA
VESPA is a neutron vibrational spectrometer designed to be used across a wide range of research areas in chemistry, physics and materials science. VESPA will access scientific domains currently out of reach for neutrons and opens new opportunities for applied research in renewable energy technologies.
Fifteen instruments are currently under construction at ESS.
The in-kind partner institutions collaborating with ESS to design and build VESPA are:
This feature is part of an ongoing Future Science series; a set of stories highlighting some of the research opportunities that will be made possible by instruments currently under construction at ESS.