Through both static and time-resolved neutron reflectometry at ISIS, a Swedish/UK research collaboration have been able to reveal the molecular mechanism of the first stage of cell death.
Programmed cell death (apoptosis) is essential for human life, and its disruption can cause cancerous cells to grow and not respond to treatment. The crucial control of the balance between cellular well-being and death is managed by a series of proteins with opposing roles from the Bcl‑2 family, which meet at the surface of mitochondria to determine a cell’s fate.
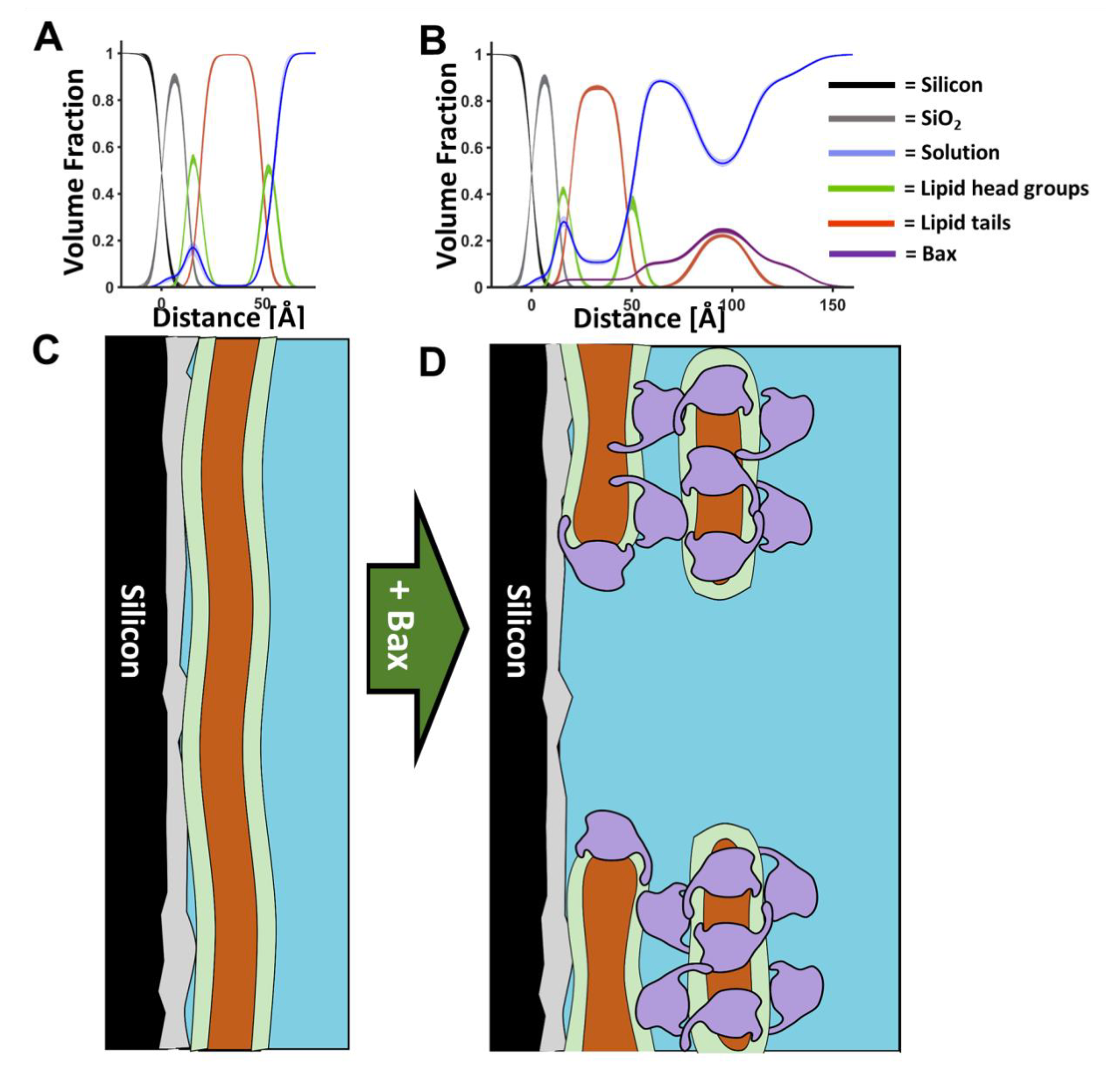
In normal cells, the soluble Bax protein is responsible for the clearance of old or diseased cells, and when activated, it perforates the mitochondrial membrane surface to form pores that trigger cell death. The pro-cell survival protein Bcl-2 is embedded within the lipid membrane, where it acts to prevent untimely cell death by capturing the cell-death promoting Bax. However, in cancer cells, Bcl-2 is often overproduced, leading to cancer cell proliferation by inhibiting Bax killing function. The precise mechanism of the pore formation by Bax and the role of the mitochondrial membrane lipids in this key cell-death initiating process have been unclear at the molecular level until now.
In this study, published in Science Advances, Professor Gerhard Gröbner and colleagues from the University of Umeå, Luke Clifton from ISIS and Hanna Wacklin-Knecht from the European Spallation Source, built on their previous studies of membrane-bound Bcl‑2, this time focusing on the mechanism of Bax. Using neutron reflectometry on the SURF and OFFSPEC beamlines at ISIS, they were able to study in real time the way that the protein interacts with lipids present in the mitochondrial membrane, during the initial stages of apoptosis. By employing deuterium-isotope labelling, they determined for the first time that when Bax creates pores, it extracts lipids from the mitochondrial membrane to form lipid-Bax clusters on the mitochondrial surface.
By using time-resolved neutron reflectometry in combination with surface infrared spectroscopy in the ISIS biolab, they were able to see that this pore creation occurred in two stages. Initial fast adsorption of Bax onto the mitochondrial membrane surface was followed by a slower formation of membrane-destroying pores and Bax-lipid clusters, which occurred simultaneously. This slower perforation process occurred on timescales of several hours, comparable to cell death in vivo.
The complex findings of this study give unique and direct evidence of the involvement of mitochondrial lipids in the structures formed during the membrane perturbing mechanism of Bax, which initiates cell death.
ISIS scientist and lead author, Luke Clifton, explains: “This work has relevance to understanding fundamental mammalian cell processes and understanding cancer biology. It really shows the capabilities of neutron reflectometry in structural studies on membrane biochemistry.”
Co-investigator Hanna Wacklin-Knecht from the European Spallation Source Deuteration and Macromolecular Crystallisation (DEMAX) Platform adds: “This collaboration with Prof. Gerhard Gröbner on cell-death regulating proteins is a great example of how neutrons can offer unique molecular-level information in biophysical and biomedical research, and in which ESS will open up new avenues to solving some of our time’s great health challenges, such as aging and cancer.”
And Gerhard Gröbner states: “The unique findings here, will not only have a significant impact in the field of apoptosis but will also open up gateways for exploring Bax and its relatives as interesting targets in cancer therapy e. g. by tuning up their cell-killing potential.
Details of this research can be found at : https://www.science.org/doi/10.1126/sciadv.adg7940.